I. ENERGY The video to the right was filmed during the loss of the Hindenburg in 1937, in which 36 people were killed. Flames consumed the zeppelin, larger than many modern ocean liners, from one end to the other within one minute, releasing a huge amount of chemical energy during the disaster. What kind of energy is being released? Can that energy be harnessed to do useful work? How do energy changes affect our everyday lives?
This unit will explore the energy changes involved in chemical reactions and how they can be measured, quantified, and predicted. The study of heat transfer in chemical reactions and physical changes (such as phase changes) is known as thermochemistry. A closely related field of chemistry is known as thermodynamics, which relates energy changes in chemical reactions with work that can be accomplished accompanying the chemical change. |
|
I.A What is Energy?

Figure I.A.1: Kinetic and Potential energy
In the study of classical physics, energy is typically defined as the ability to do work. There are two kinds of energy with which you are probably familiar: potential energy (stored energy of position) and kinetic energy (energy of motion). In Figure II.A, potential energy can be envisioned as the energy stored in the roller-coaster car at the top of the track, and kinetic energy would be energy of motion in the car as it speeds down the track and zooms up the next hill. Kinetic and potential energy can easily be translated from one form to the other and back again.
In chemistry, chemical potential energy is stored in chemical bonds and released in the form of heat. Chemical potential energy is stored in the type of bonds, the molecular motion, and the composition of the material. Kinetic energy in chemistry is observed in the form of heat energy released when bonds are formed, or absorbed when bonds are broken. During a chemical reaction, it takes energy input to break chemical bonds, and energy is released when new bonds are formed.
There are some exceptions to this general rule, such as chemiluminescent reactions that release light energy instead of heat during a chemical reaction. A common example of chemiluminescence is a glowstick. called triboluminescence (see sidebar below). On the whole, however, we will measure energy changes in chemical reactions in the form of heat.
In chemistry, chemical potential energy is stored in chemical bonds and released in the form of heat. Chemical potential energy is stored in the type of bonds, the molecular motion, and the composition of the material. Kinetic energy in chemistry is observed in the form of heat energy released when bonds are formed, or absorbed when bonds are broken. During a chemical reaction, it takes energy input to break chemical bonds, and energy is released when new bonds are formed.
There are some exceptions to this general rule, such as chemiluminescent reactions that release light energy instead of heat during a chemical reaction. A common example of chemiluminescence is a glowstick. called triboluminescence (see sidebar below). On the whole, however, we will measure energy changes in chemical reactions in the form of heat.
Sidebar: Triboluminescence
|
Light can also be produced as a result of physical stress. A good example of this is called triboluminescence, which can be observed when a Wintergeen Lifesaver candy is crushed in a very dark room. While triboluminescence is not thoroughly understood, it is thought to occur because of a brief separation of positive and negative charges when certain types of crystals (sugar in particular) are fractured. An electric potential is created across the gap of the fractured sugar crystal, and electrons will jump across the gap. In the process, they ionize other electrons in nitrogen gas in the atmosphere, which emit ultraviolet radiation, causing the wintergreen flavoring (methyl salicylate) to fluoresce. Triboluminescence is also observed in some when opening certain band-aid wrappers and scotch tape. Try it!
|
I.B Conservation of Energy
When the roller coaster car zooms down the hill, the stored potential energy of position is converted to kinetic energy of motion. When natural gas (methane, CH4) burns in your house’s heating unit, some of the chemical potential energy of the gas, stored within the chemical bonds of the methane, is converted to heat. In both examples, energy changes from one form to another. Is there a loss of total energy?
The answer is NO. While energy is converted from one form to another, the total amount of energy remains constant. Energy is said to be conserved. This can be restated as the law of conservation of energy, otherwise known as the First Law of Thermodynamics: in any chemical reaction or physical process, energy can be converted from one form to another but is never created nor destroyed.
In chemistry, a large amount of energy may be released by a chemical reaction. However, that energy must go somewhere and can be completely accounted for in controlled situations. Therefore, the total energy lost by the chemicals in a reaction is conserved.
The answer is NO. While energy is converted from one form to another, the total amount of energy remains constant. Energy is said to be conserved. This can be restated as the law of conservation of energy, otherwise known as the First Law of Thermodynamics: in any chemical reaction or physical process, energy can be converted from one form to another but is never created nor destroyed.
In chemistry, a large amount of energy may be released by a chemical reaction. However, that energy must go somewhere and can be completely accounted for in controlled situations. Therefore, the total energy lost by the chemicals in a reaction is conserved.
I.C Measuring Heat
In chemistry, we can measure heat changes using temperature, which is simply a measure of the average kinetic energy of the particles in a sample of matter. This is intrinsically different from heat, however, which can be thought of as the energy transferred between samples of matter due to a difference in their temperature. Temperature does not depend on the amount of material present and is sometimes called an intensive property, whereas heat is an extensive property, which depends on the amount of the sample that is present.
|
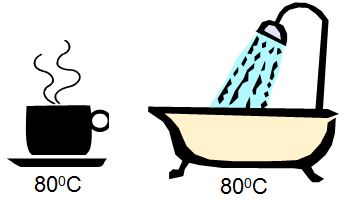
For example, two containers full of water (a coffee cup and a bathtub; Fig. I.C.1) can be heated to the same temperature. If heat is released from both at the same rate, which sample would cool to room temperature more quickly? The amount of heat transferred by the samples depends on the amount of sample present, an extensive property; the amount of sample has no bearing on the initial or final temperatures, an intensive property.
Mass and density are other examples of extensive and intensive properties. A sample of gold will have the same density (19.3 g/cm3) regardless of the size of the sample. As the sample grows in mass, so will the volume, and the ratio of mass to volume (or M/V) remains the same. Density is said to be an intensive property, whereas mass is an extensive property. |
|
In chemistry we measure heat in calories (cal), which is defined as the amount of heat required to raise one gram of water one degree Celsius. You have probably heard of nutritional Calories, as stated on the back of most food products. However, these Calories are NOT the same as a chemistry calorie. Note that a nutritional Calorie is denoted with a capital C, whereas a chemistry calorie is denoted with a small c. A nutritional Calorie is equal to 1,000 chemistry calories and is also known as a kilocalorie (kcal). Therefore, 1 kcal = 1 Calorie. Easy!
The SI unit of heat and energy is the joule (J). One joule is equal to 0.239 calories; one calorie is therefore equal to 4.184 joules. Example Problem: When an Oreo cookie is combusted, 55 nutritional Calories are released. How much heat is released in joules?
55 Calories x 1,000 cal/Calorie x 4.184 J/cal = 230,000 J |
I.D Specific Heat
A calorie is the amount of heat needed to raise one gram (equal to 1 mL) of water 1 oC. This is also known as the specific heat of water, which can also be expressed as 4.184 joules per gram per degree Celsius (J/[g∙ oC]).
Eqn. 1: s(water) = 1 cal = 4.184 J/(g∙ oC)
The specific heat (s) of any substance is defined as the amount of heat needed to raise one gram of a substance one degree Celsius.
Eqn. 1: s(water) = 1 cal = 4.184 J/(g∙ oC)
The specific heat (s) of any substance is defined as the amount of heat needed to raise one gram of a substance one degree Celsius.
|
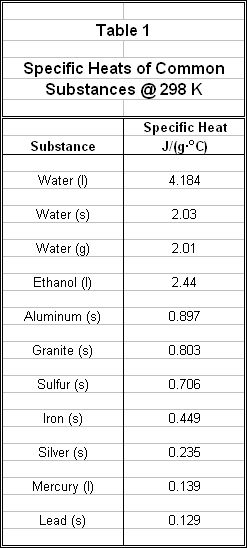
Specific heat is an extensive and characteristic property of matter, meaning that its value is unique and can be used to identify a particular type of substance. For example, the specific heats of some different substances are listed in Table 1. Note the unusually high specific heat of liquid water. This means that it takes more energy in calories to heat a specific mass of water than it would take to raise the same mass of (for example) lead the same number of degrees C. In other words, water stores significantly more heat energy per degree of Celsius change than lead, or almost any other form of matter. This has deep implications for life on Earth, as the planet resists big changes in temperature because water, which covers 2/3 of the Earth’s surface, acts as a heat sink in warmer climates and a heat source in cooler climates.
The amount of heat it takes to change the temperature of a substance depends on three factors: the nature of the material itself (i.e., its specific heat, a characteristic property), the amount or mass (m) of the material that is being heated, and the total change in temperature (ΔT). For example, it takes much more heat (in cal) to warm a swimming pool to comfortable temperatures than it would to warm a glass of water over the same ΔT. We can express these relationships in the following equation: Eqn. 2: q = s ∙ m ∙ ΔT where q = heat absorbed or released (J); s = specific heat of substance (J/[g∙ oC]); m = mass of sample (g); and ΔT = difference between final and initial temperature (oC), or Tfinal – Tinitial = ΔT. We can use this formula to calculate the amount of heat necessary to warm a sample of matter, say water. As an example, what amount of heat (in calories) would be needed to heat an 8-ounce cup of coffee from room temperature (about 21 oC) to 80 oC? |
First, we need to convert the volume of water (8 fluid ounces) to mass of water in SI units. One fluid ounce is equal to 29.6 mL, so:
8 fluid ounces ∙ 29.6 mL/1 fluid ounce ∙ 1 g/mL = 237 g
We also know the following:
s(H2O) = 4.184 J/g∙ oC ΔT = Tfinal – Tinitial = 80 oC – 21 oC = 59 oC
We can substitute into the equation as follows:
q = s ∙ m ∙ ΔT
q = (4.184 J/ g∙ oC) ∙ (237 g) ∙ (59 oC)
q = 58,500 J
We can convert this result to calories as follows:
58,500 J ∙ 1 cal/4.184 J = 14,000 cal
8 fluid ounces ∙ 29.6 mL/1 fluid ounce ∙ 1 g/mL = 237 g
We also know the following:
s(H2O) = 4.184 J/g∙ oC ΔT = Tfinal – Tinitial = 80 oC – 21 oC = 59 oC
We can substitute into the equation as follows:
q = s ∙ m ∙ ΔT
q = (4.184 J/ g∙ oC) ∙ (237 g) ∙ (59 oC)
q = 58,500 J
We can convert this result to calories as follows:
58,500 J ∙ 1 cal/4.184 J = 14,000 cal
Example ProblemA sample of granite warms in the sunlight from its morning temperature (16.0 oC) to its maximum temperature (78.0 oC) by absorbing 143 kJ of heat. What is the mass of the sample of granite? This problem is solved in the Voicethread on the right, or click here to go directly to the VoiceThread site:
|
|
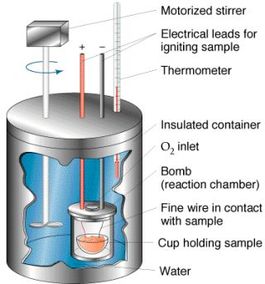
I.E Calorimetry The vast majority of chemical reactions will include some sort of change in energy, typically heat. Heat changes in reactions are measured accurately in the lab using an instrument known as a calorimeter . Calorimeters are insulated vessels that measure heat absorbed or released during a chemical reaction. Some instruments are called “bomb” calorimeters (Fig. I.E.1) because they are tightly sealed off from the external environment and are used specifically to measure the heats of combustion reactions.
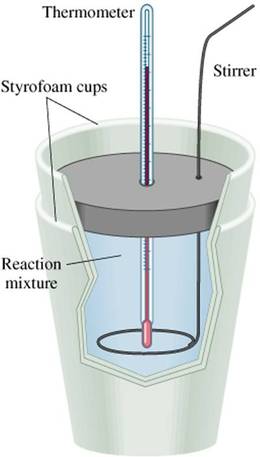
Bomb calorimeters are precision instruments that are expensive to purchase and maintain. However, reasonable results for high-school chemistry experiments can be gleaned from a coffee-cup calorimeter, as shown in Figure I.E.2. Because the coffee-cup calorimeter is not tightly sealed, the vessel will, in effect, be open to the atmosphere and any experiments will therefore occur at constant pressure. These are sometimes called "constant pressure" calorimeters. The data that is collected in calorimetry is the temperature change measured in a known volume of water within the calorimeter. Because both the specific heat (4.184 J/g∙oC) and the volume of water in the calorimeter are known values, and the ΔT is measured, the value for q (heat of reaction) can be solved for using Eqn. 2. Calorimeters are often used to determine the energy content of food products. The energy released during the combustion of glucose in a calorimeter is analogous to the energy available to your cells during the metabolic respiration of the food you eat. Cellular respiration is essentially a combustion reaction of glucose, mediated by certain catalysts and proteins that change the rate of the combustion reaction: C6H12O6(s) + 6O2(g) --> 6CO2(g) + 6H2O(l) Therefore, a lab calorimeter can be used to determine the energy content of food products. Example Problem Suppose a calorimeter containing 400 mL of water is used to determine the caloric content of a potato chip with a mass of 2.83 grams. When the chip is combusted in the calorimeter, the water in the calorimeter is heated from room temperature (21.0 oC) to 59.8 oC. How many nutritional calories are in the potato chip?
This problem is solved in the VoiceThread shown below, or click here to view the VoiceThread directly. |
Calorimetry can also be used to calculate the specific heat of an unknown substance, which can later be compared to accepted and published values. Remember that specific heat is a characteristic property of matter, so the experimental value of specific heat determined in the lab can be used to identify an unknown.
Example Problem A coffee-cup calorimeter contains 125 mL of water at an initial temperature of 25.6 oC. A 50.0g sample of unknown metal is placed in a boiling water bath and allowed to equilibrate to the boiling temperature of water. When the unknown metal is removed and placed into the calorimeter, the final equilibrium temperature of the metal and water in the calorimeter is 28.7 oC. Can you calculate the specific heat of the unknown metal? By comparing the calculated value to Table 1, can you identify the unknown metal?
|
|
The key to solving the previous example problem is the first law of thermodynamics, which states that energy is not created nor destroyed during a chemical or physical change. Assuming no heat is lost to the surroundings, the heat gained by the water in the calorimeter must be equal to the heat lost by the metal sample. We can therefore calculate the heat gained by the water in the calorimeter (q), then set that value equal to the heat lost by the unknown metal, after which we can solve for the specific heat (s) of the metal. This problem is solved in the VoiceThread shown above, or you can click here to view it directly.