III. CALCULATING ENTHALPIES OF REACTION
III.A Hess's Law
While a calorimeter can be used to measure the ΔH of many chemical changes, some reactions are not easy to duplicate in a lab because they proceed too slowly or may not create the desired product. For reactions like this, chemists use a theoretical way to calculate ΔH.
Hess’s Law of heat summation provides a method to calculate the enthalpy of any reaction not included in the published data tables. Hess’s law states that the total enthalpy change for a multi-step reaction is equal to the sum of the enthalpy changes for each individual step. In other words, heat changes in reactions are additive if the overall reaction has more than one step.
As an example, let’s try to calculate the ΔH for the synthesis of methane gas from solid carbon (graphite) and hydrogen gas:
C(s) + 2H2(g) --> CH4(g) ΔHrxn = ?
We can use the published values for the individual combustion reactions of carbon, hydrogen gas, and methane to solve this problem:
C(s) + O2(g) --> CO2(g) ΔHcomb = -394 kJ
H2(g) + ½O2(g) --> H2O(l) ΔHcomb = -286 kJ
CH4(g) + 2O2(g) --> CO2(g) + 2H2O(l) ΔHcomb = -891 kJ
The reactions can not be summed in their given configurations to result in the equation for the synthesis of methane. However, we can manipulate the equations and then combine them to get the reaction equations needed to correctly apply Hess’s law. Two general principles of manipulating thermochemical equations must be clearly understood before we begin: (1) If a reaction is reversed, the sign for ΔH is also reversed; and (2) if a thermochemical equation is multiplied, ΔH must also be multiplied by the same factor.
Because we want methane to be a product in the final reaction, we reverse the combustion equation for methane:
CO2(g) + 2H2O(l) --> CH4(g) + 2O2(g) ΔHcomb = +891 kJ
In this reversed reaction, two moles of water are used as a reactant. Therefore we must multiply the combustion reaction for hydrogen gas (which has water as a product) by two to obtain the desired amount of water:
2H2(g) + O2(g) --> 2H2O(l) ΔHcomb = -572 kJ
We are now able to add the three modified reactions together, using Hess’s law, to calculate the total enthalpy of the reaction for the formation of methane from its elements:
C(s) + O2(g) --> CO2(g) ΔHcomb = -394 kJ
2H2(g) + O2(g) --> 2H2O(l) ΔHcomb = -572 kJ
CO2(g) + 2H2O(l) --> CH4(g) + 2O2(g) ΔHcomb = +891 kJ
____________________________________________________
C(s) + 2H2(g) --> CH4(g) ΔHrxn = -75 kJ
Hess’s Law states that the enthalpy difference across a reaction is independent of the path the reaction takes toward the ultimate products. If no data existed for the ΔHrxn for the synthesis of methane, we can still construct a three-step reaction that, when manipulated and summed, accurately approximates the ΔHrxn for this reaction.
Hess’s Law of heat summation provides a method to calculate the enthalpy of any reaction not included in the published data tables. Hess’s law states that the total enthalpy change for a multi-step reaction is equal to the sum of the enthalpy changes for each individual step. In other words, heat changes in reactions are additive if the overall reaction has more than one step.
As an example, let’s try to calculate the ΔH for the synthesis of methane gas from solid carbon (graphite) and hydrogen gas:
C(s) + 2H2(g) --> CH4(g) ΔHrxn = ?
We can use the published values for the individual combustion reactions of carbon, hydrogen gas, and methane to solve this problem:
C(s) + O2(g) --> CO2(g) ΔHcomb = -394 kJ
H2(g) + ½O2(g) --> H2O(l) ΔHcomb = -286 kJ
CH4(g) + 2O2(g) --> CO2(g) + 2H2O(l) ΔHcomb = -891 kJ
The reactions can not be summed in their given configurations to result in the equation for the synthesis of methane. However, we can manipulate the equations and then combine them to get the reaction equations needed to correctly apply Hess’s law. Two general principles of manipulating thermochemical equations must be clearly understood before we begin: (1) If a reaction is reversed, the sign for ΔH is also reversed; and (2) if a thermochemical equation is multiplied, ΔH must also be multiplied by the same factor.
Because we want methane to be a product in the final reaction, we reverse the combustion equation for methane:
CO2(g) + 2H2O(l) --> CH4(g) + 2O2(g) ΔHcomb = +891 kJ
In this reversed reaction, two moles of water are used as a reactant. Therefore we must multiply the combustion reaction for hydrogen gas (which has water as a product) by two to obtain the desired amount of water:
2H2(g) + O2(g) --> 2H2O(l) ΔHcomb = -572 kJ
We are now able to add the three modified reactions together, using Hess’s law, to calculate the total enthalpy of the reaction for the formation of methane from its elements:
C(s) + O2(g) --> CO2(g) ΔHcomb = -394 kJ
2H2(g) + O2(g) --> 2H2O(l) ΔHcomb = -572 kJ
CO2(g) + 2H2O(l) --> CH4(g) + 2O2(g) ΔHcomb = +891 kJ
____________________________________________________
C(s) + 2H2(g) --> CH4(g) ΔHrxn = -75 kJ
Hess’s Law states that the enthalpy difference across a reaction is independent of the path the reaction takes toward the ultimate products. If no data existed for the ΔHrxn for the synthesis of methane, we can still construct a three-step reaction that, when manipulated and summed, accurately approximates the ΔHrxn for this reaction.
|
EXAMPLE PROBLEM: Hydrogen peroxide (H2O2) is a common substance used in many applications, from dying hair to providing an oxidant for jet engines (including jetpacks, see video). H2O2 decomposes spontaneously according to the following reaction:
2H2O2(l) --> 2H2O(l) + O2(g) Use the following two thermochemical equations to solve ΔHrxn for the decomposition of hydrogen peroxide. 2H2(g) + O2(g) --> 2H2O(l) ΔHcomb = -572 kJ H2(g) + O2(g) --> H2O2(l) ΔHcomb = -188 kJ This problem is solved in the VoiceThread shown to the right, or you can click here to view directly. |
|
III.B Enthalpy of Formation
We have observed that the total enthalpy of a reaction can be estimated using published experimental values for the enthalpies of several combustion reactions. However, tabulating the enthalpy changes for every possible combustion reaction in the universe would be a huge task, so scientists have identified a simpler method to estimate the ΔH of a reaction. To do this, chemists have recorded the enthalpy change for a particular type of reaction, a synthesis reaction in which the compound of interest is formed from its constituent elements in their standard states (1 atm and 298K).
The standard enthalpy (heat) of formation (ΔHof) is defined as the heat change that occurs when one mole of a compound is formed from its constituent elements in the standard state. For example, the standard enthalpy of formation for one mole of water is written as shown below:
H2(g) + ½O2(g) --> H2O(l) ΔHof = -286 kJ
The elements oxygen and hydrogen are diatomic in their standard states, and water is a liquid in its standard state. Because ΔHof is always computed per mole of product, there is only one mole of water listed as a product, and the coefficient for oxygen must therefore be one half. Most (but not all) enthalpies of formation are negative, meaning that most synthesis reactions of compounds from their pure elements are exothermic (for example, the synthesis of methane from carbon and hydrogen gas shown in Section III.A).
We have observed that the total enthalpy of a reaction can be estimated using published experimental values for the enthalpies of several combustion reactions. However, tabulating the enthalpy changes for every possible combustion reaction in the universe would be a huge task, so scientists have identified a simpler method to estimate the ΔH of a reaction. To do this, chemists have recorded the enthalpy change for a particular type of reaction, a synthesis reaction in which the compound of interest is formed from its constituent elements in their standard states (1 atm and 298K).
The standard enthalpy (heat) of formation (ΔHof) is defined as the heat change that occurs when one mole of a compound is formed from its constituent elements in the standard state. For example, the standard enthalpy of formation for one mole of water is written as shown below:
H2(g) + ½O2(g) --> H2O(l) ΔHof = -286 kJ
The elements oxygen and hydrogen are diatomic in their standard states, and water is a liquid in its standard state. Because ΔHof is always computed per mole of product, there is only one mole of water listed as a product, and the coefficient for oxygen must therefore be one half. Most (but not all) enthalpies of formation are negative, meaning that most synthesis reactions of compounds from their pure elements are exothermic (for example, the synthesis of methane from carbon and hydrogen gas shown in Section III.A).
|
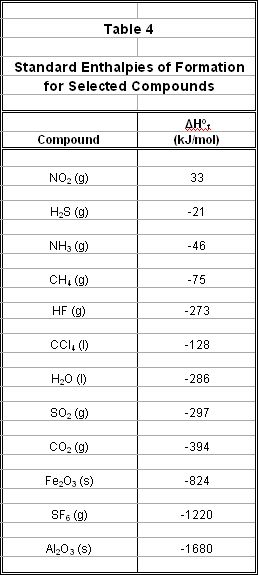
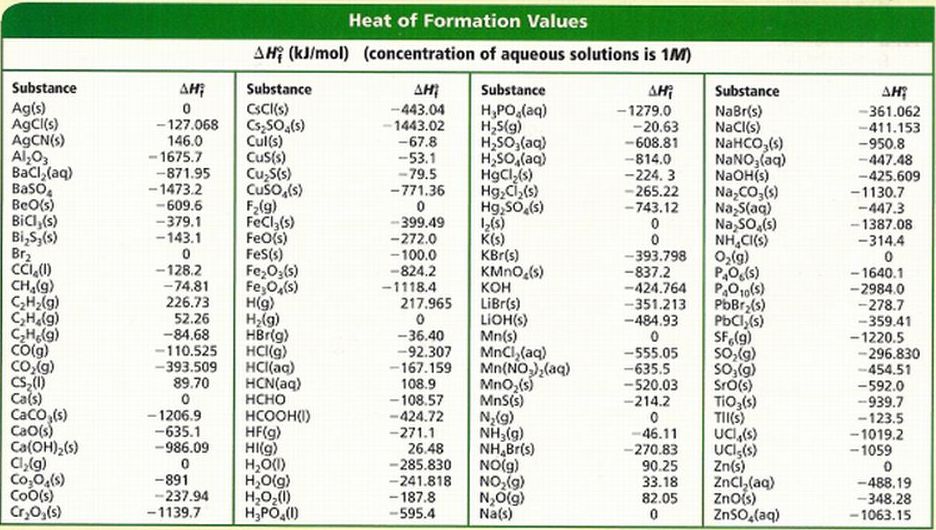
It is important to recognize that the standard enthalpy of formation for a pure element in its standard state is zero. A short list of tabulated values for the ΔHof for some reactions is included in Table 4 and a longer list can be referenced at the bottom of this page.
Can we use the tabulated values of ΔHof to approximate the enthalpy of any reaction? Using an extension of Hess’s Law, we can add the values of ΔHof for the products and reactants in any reaction to approximate the overall ΔHorxn, as follows: Eqn. 5: ΔHorxn = ΣΔHof(products) - ΣΔHof(reactants) The symbol Σ means “the sum of.” The formula above states that the enthalpy change for any reaction can be calculated by subtracting the sum of the standard enthalpies of formation of the reactants from the sum of the standard enthalpies of formation of the products. Example Problem: The industrial production of iron is accomplished in a blast furnace by reacting an ore of iron oxide (Fe2O3) with carbon, according to the following balanced chemical reaction:
2Fe2O3(s) + 3C(s) → 4Fe(s) + 3CO2(g) Calculate the ΔHorxn for the production of iron using the tabulated values of ΔHof (see Table 4). ΔHorxn = [(4)ΔHof(Fe) + (3)ΔHof(CO2)] – [(3)ΔHof(C) + (2)ΔHof(Fe2O3)] ΔHorxn = [(4)(0) + (3)(-394 kJ)] – [(3)(0) + (2)(-824 kJ)] ΔHorxn = [-1,182 kJ] – [-1,648 kJ] ΔHorxn = + 466 kJ |
Because the sign for ΔHorxn is positive, the production of iron is an endothermic reaction (which makes sense, given that it requires a high-temperature blast furnace for the reaction to proceed). Note that all heats of formation are multiplied by their respective molar coefficients from the balanced equation, and that the ΔHof of the pure elements carbon and iron are zero.
Due to a number of complicated interactions other than simple bond formation and destruction, the calculation of ΔHorxn does not produce perfectly accurate results. Experimental values of ΔHorxn are always somewhat different from calculated values. However, a reasonably good approximation of ΔHorxn can be obtained using this method, and we can certainly assess whether a reaction is exothermic or endothermic using this method.
Due to a number of complicated interactions other than simple bond formation and destruction, the calculation of ΔHorxn does not produce perfectly accurate results. Experimental values of ΔHorxn are always somewhat different from calculated values. However, a reasonably good approximation of ΔHorxn can be obtained using this method, and we can certainly assess whether a reaction is exothermic or endothermic using this method.
|
Example Problem: Using the tabulated values for the ΔHof, calculate the ΔHorxn for the combustion of ammonia gas:
4NH3(g) + 7O2(g) --> 4NO2(g) + 6H2O(l) This problem is solved in the Voicethread shown to the right, or click here to view directly. |
|