IV. THE DRIVING FORCE OF REACTIONS
IV.A Reaction Spontaneity When an iron object is left outdoors in moist air, a spontaneous chemical reaction occurs:
4Fe(s) + 3O2(g) --> 2Fe2O3(s) ΔHrxn = -1,625 kJ This highly exothermic reaction is also used in certain commercial hand-warmers. The “rust” reaction proceeds in the forward direction without any input form outside forces and is considered a spontaneous chemical reaction. A spontaneous process is any physical or chemical change that occurs without intervention from outside the system. However, many spontaneous chemical reactions require some activation energy to initiate the process, although the process may |
continue spontaneously following the removal of an energy input. For example, the combustion of natural gas (methane) to heat your home requires a pilot light or spark to start the reaction, although the combustion proceeds spontaneously thereafter. The majority of spontaneous chemical reactions in nature are exothermic. Products of exothermic reactions are in a lower potential energy state and are therefore more stable and less reactive than the original reactants. We might think that endothermic reactions, in which energy is absorbed, can not be spontaneous without a continued energy input. However, this conclusion is incorrect, and it is not hard to find an example of a spontaneous endothermic process:
H2O(s) --> H2O(l) ΔH = +6.0 kJ
This reaction depicts the melting of ice under standard conditions. We can safely conclude that some factor other than enthalpy change plays a role in determining spontaneity. That factor is called entropy.
Entropy (S) is a measure of the degree of randomness, or disorder, in the particles in a system. The melting of ice increases the entropy of the system by changing the arrangement of water molecules from a highly ordered crystalline solid to a disordered liquid with a much greater degree of molecular movement. Why is it that the appetizing smell of baking cookies doesn’t stay in the kitchen, or that coffee creamer will mix fully with coffee without stirring? In both of these examples, there is neither an energy loss (i.e., they are not exothermic), nor is there any intentional mixing of the reactants, yet the process occurs. The reason is entropy, a spontaneous increase in the disorder of the systems.
The tendency toward disorder is one of the driving forces of physical and chemical processes. This is summarized in the second law of thermodynamics, which states that in spontaneous processes, the entropy (disorder) in the universe always increases.
H2O(s) --> H2O(l) ΔH = +6.0 kJ
This reaction depicts the melting of ice under standard conditions. We can safely conclude that some factor other than enthalpy change plays a role in determining spontaneity. That factor is called entropy.
Entropy (S) is a measure of the degree of randomness, or disorder, in the particles in a system. The melting of ice increases the entropy of the system by changing the arrangement of water molecules from a highly ordered crystalline solid to a disordered liquid with a much greater degree of molecular movement. Why is it that the appetizing smell of baking cookies doesn’t stay in the kitchen, or that coffee creamer will mix fully with coffee without stirring? In both of these examples, there is neither an energy loss (i.e., they are not exothermic), nor is there any intentional mixing of the reactants, yet the process occurs. The reason is entropy, a spontaneous increase in the disorder of the systems.
The tendency toward disorder is one of the driving forces of physical and chemical processes. This is summarized in the second law of thermodynamics, which states that in spontaneous processes, the entropy (disorder) in the universe always increases.
IV.B Quantifying Entropy
Calculation of the value of entropy (S) is complicated and difficult, but it can be done. Most chemical reference texts will include tabulated (experimental) values of the entropy of a reaction. A determination of the sign for S is probably more valuable for our purposes.
The change in entropy (ΔS) across a reaction is calculated in much the same way as ΔHrxn:
ΔSsystem = ΔSproducts - ΔSreactants
If the entropy of a system increases during a reaction, then ΔSproducts > ΔSreactants and the value for ΔSsystem is positive, and vice versa.
There are a number of general rules for predicting whether ΔSsystem is positive or negative across a reaction.
1. If the number of gas particles increases across a reaction, entropy generally increases and ΔSsystem is positive. In the following reaction, the sign for ΔSsystem is negative because three molecules of gas react to produce just two molecules of another gas:
2CO(g) + O2(g) --> 2CO2(g) ΔSsystem is negative (< 0)
2. Dissolving a solid or liquid always increases the entropy of a system, because dissolved particles always have more chaotic and random molecular motion than within a pure state:
CaCl2(s) --> Ca2+(aq) + 2Cl-(aq) ΔSsystem is positive (> 0)
3. Dissolving of a gas always results in a decrease in entropy, because the particles will have much less freedom of motion in the aqueous state than in the gaseous state:
CO2(g) --> CO2(aq) ΔSsystem is negative (< 0)
4. Changes of state are associated with entropy changes . Endothermic changes of state, such as melting or vaporization, will have a positive ΔSsystem, whereas exothermic phase changes, such as freezing or condensation, will have a negative ΔSsystem.
5. An increase in temperature is always associated with an increase in entropy, simply because the average kinetic energy of the molecules (and therefore the number of possible arrangements in space) has increased.
The change in entropy (ΔS) across a reaction is calculated in much the same way as ΔHrxn:
ΔSsystem = ΔSproducts - ΔSreactants
If the entropy of a system increases during a reaction, then ΔSproducts > ΔSreactants and the value for ΔSsystem is positive, and vice versa.
There are a number of general rules for predicting whether ΔSsystem is positive or negative across a reaction.
1. If the number of gas particles increases across a reaction, entropy generally increases and ΔSsystem is positive. In the following reaction, the sign for ΔSsystem is negative because three molecules of gas react to produce just two molecules of another gas:
2CO(g) + O2(g) --> 2CO2(g) ΔSsystem is negative (< 0)
2. Dissolving a solid or liquid always increases the entropy of a system, because dissolved particles always have more chaotic and random molecular motion than within a pure state:
CaCl2(s) --> Ca2+(aq) + 2Cl-(aq) ΔSsystem is positive (> 0)
3. Dissolving of a gas always results in a decrease in entropy, because the particles will have much less freedom of motion in the aqueous state than in the gaseous state:
CO2(g) --> CO2(aq) ΔSsystem is negative (< 0)
4. Changes of state are associated with entropy changes . Endothermic changes of state, such as melting or vaporization, will have a positive ΔSsystem, whereas exothermic phase changes, such as freezing or condensation, will have a negative ΔSsystem.
5. An increase in temperature is always associated with an increase in entropy, simply because the average kinetic energy of the molecules (and therefore the number of possible arrangements in space) has increased.
Example Problems: Predict the sign of ΔSsystem for each of the following chemical reactions:
(a) CH3OH(l) --> CH3OH(g) ΔSsystem is positive (> 0)
(b) Mg(NO3)2(s) --> Mg2+(aq) + 2NO3-(aq) ΔSsystem is positive (> 0)
(c) ClF(g) + F2(g) --> ClF3(g) ΔSsystem is negative (< 0)
(a) CH3OH(l) --> CH3OH(g) ΔSsystem is positive (> 0)
(b) Mg(NO3)2(s) --> Mg2+(aq) + 2NO3-(aq) ΔSsystem is positive (> 0)
(c) ClF(g) + F2(g) --> ClF3(g) ΔSsystem is negative (< 0)
IV.C Free Energy
We have learned that processes in nature are driven in two directions: toward lower enthalpy (exothermia) and toward greater entropy (disorder). Using this information, is it possible to predict whether a reaction is spontaneous?
|
In 1878, Yale University physicist J. Willard Gibbs (Fig IV.C.1) defined a function that combines enthalpy and entropy into one term that helps predict reaction spontaneity, called Gibbs free energy (ΔGsyst). Gibbs free energy, commonly called the free energy of a system, is the energy that is available to do work after a chemical reaction, and is thus considered useful energy. Unfortunately, some of the energy of a chemical reaction goes into non-useful functions, such as random molecular motion (i.e., entropy), which can not be recovered to do useful work.
The standard Gibbs free energy change in a reaction (ΔGsyst) is therefore defined as the enthalpy change of a reaction (ΔHsyst) minus the entropy change (ΔSsyst), which depends on the temperature (in degrees Kelvin): Eqn. 5: ΔGosyst = ΔHosyst - TΔSosyst The sign for ΔGosyst determines whether a reaction is spontaneous. Highly spontaneous reactions include those that lower the energy (i.e., are exothermic) and increase the entropy of the system. In this case, ΔHosyst is negative and ΔSosyst is positive. Substituting these |
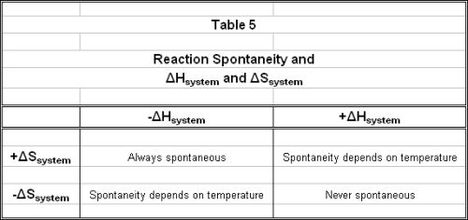
values in to Eqn. 6 would result in a negative number. Therefore, if ΔGosyst is negative, the process is spontaneous and will proceed without external influence. However, if ΔGosyst is positive, the reaction is non-spontaneous and energy must be continually input to the system for the reaction to proceed. Table 5 relates the signs of ΔHosyst and ΔSosyst to reaction spontaneity.
|
Example Problem: The production of ammonia is simply a reaction between nitrogen and hydrogen gases:
N2(g) + 3H2(g) --> 2NH3(g) ΔHosyst = -92 kJ ; ΔSosyst = -197 J/K While this is an exothermic process, the entropy of the system decreases significantly as four gas molecules of the reactants are converted to two gaseous product molecules. Which of the two conditions will dominate the process, and is the reaction spontaneous under standard conditions? Assume the reaction proceeds under standard conditions. This problem is solved in the VoiceThread shown to the right, or you can click here to view Voicethread icon to view. |
|